Chi Van Dang generally declines to discuss the science that made him famous. A leading authority on cancer metabolism, he routinely is asked to speak about how tumors reprogram biochemical pathways to help them slurp up nutrients, and how disrupting these noxious adaptations could be a powerful approach to treating cancer.
Instead, Dang uses his soapbox at every research meeting, every invited lecture and every blue-ribbon panel to advocate for something else entirely: a simple yet radical tweak to how oncologists administer cancer drugs.
The approach, known as chronotherapy, involves timing the delivery of drugs so as to minimize the side effects of treatment while maximizing the treatment’s effectiveness. The idea is to synchronize therapy with the body’s natural 24-hour rhythms — the so-called circadian clock — striking either when cancer cells are most vulnerable to assault or when healthy cells are least sensitive to toxicity (or, ideally, both).
“It’s just always been on the fringe. There weren’t that many card-carrying cancer biologists like me getting into it.”
Chi Van DangDang didn’t set out to become a global ambassador for this field. But as scientific director of the Ludwig Institute for Cancer Research, a nonprofit that funds hundreds of cancer labs worldwide, and chair of the board of scientific advisors at the US National Cancer Institute, he finds himself in a powerful position to reshape the research agenda — and he believes chronotherapy’s time has come.
It is not an entirely new concept. The idea of time-stipulated therapy dates back decades, with some randomized trials in the 1980s and 1990s showing dramatic reductions in toxicities and extended survival times among cancer patients treated in a clock-optimized fashion. But for the most part, “it’s just always been on the fringe,” says Dang. “There weren’t that many card-carrying cancer biologists like me getting into it.”
Until now.
The concept of chronotherapy got a major boost last year when the Nobel Prize in Physiology or Medicine went to three biologists who first uncovered the inner workings of our bodies’ daily rhythms. Scientists who study the interface of circadian biology and cancer say there’s been an uptick in interest in their work since the prize was announced last October.
But arguably, Dang’s own efforts to spread the gospel about the cancer-clock connection and its therapeutic implications have been even more important in revitalizing the field. “It’s capturing people’s interest,” he says — with a growing number of cancer researchers now beginning to explore the ways in which circadian rhythms shape tumor development.
They are finding not only new ways of administering old drugs but also clever tactics for rewiring aberrant clocks. And they are transforming a therapeutic strategy long dismissed as complementary or alternative medicine into a rigorous science.
“Time is an inconvenient truth” — one that oncologists and cancer researchers alike, Dang says, can no longer afford to ignore.
Timing is of the essence
A slender and bespectacled 63-year-old with the confident and unhurried voice of a seasoned physician, Dang cites his father — Chieu Van Dang, Vietnam’s first neurosurgeon and former dean of the University of Saigon School of Medicine — as a role model for how he approaches positions of leadership in academic medicine. His father’s death from liver cancer in 2004 remains a lasting inspiration to develop better treatments. And family, again, was a driving factor behind a fortuitous career move that prompted Dang’s recent zeal for circadian biology.
He had spent nearly 25 years on faculty at Johns Hopkins University, during which he rose through the ranks to become vice dean for research of the medical school; he figured he’d never leave. But in 2011, after his older brother Bob died of metastatic cancer of the soft tissues, Dang thought to himself: “As a medical oncologist and researcher, I need to do more.” So when the University of Pennsylvania approached him with an offer to become director of its cancer center — home to the original discovery that abnormal chromosomes can cause cancer, and where a new generation of lifesaving T-cell therapies were being developed — he jumped at the opportunity.
As luck would have it, Penn also is home to one of the largest assemblages of chronobiology researchers in the country, and Dang soon found himself chatting with — and then collaborating with — clock researchers from across the Philadelphia campus. Those interactions, he says, prompted an academic epiphany: If cancer is a disease of runaway cellular growth, and circadian rhythms are what normally keep the cell cycle in check, then disruption of the internal clock must be, as Dang puts it, a “missing link” of tumor development and growth.

Researcher Chi Van Dang is convinced that cancer treatments will be more effective and less toxic if we factor in the body's circadian rhythms.
CREDIT: THE WISTAR INSTITUTE
The circadian clock is a complex biological circuit that controls the daily rhythms of sleep, eating habits, body temperature and many other important physiological functions. The body has a master clock in the brain and many secondary clocks in other organs, as well as individual clocks in each and every cell, all controlled by an intricate network of oscillating genes and proteins.
When the various clocks are in sync, the body operates like a well-oiled machine. But when certain clock genes are mutated or thrown out of alignment by chronic jet lag, these systems can get out of whack, creating prime conditions for tumors to grow and spread.
“If I was still in Baltimore, I would probably be working on something else,” says Dang (who last year moved his lab again, this time just half a mile away to the Wistar Institute, an independent research center on the Penn campus, where he is a professor in the molecular and cellular oncogenesis program). And by extension, if Dang hadn’t started extolling the virtues of this research to officials at the National Cancer Institute, it’s unlikely that the agency — the largest funder of cancer research in the world — would ever have lined up behind the idea. It did so last year, putting out a call for grant applications from scientists seeking to better understand how circadian processes affect tumor development and responses of patients to therapy.
According to Dan Xi, a program director at the NCI, there’s now talk of creating a “human circadian rhythm atlas” to bring systems-level understanding to the study of clock disruption in cancer, immunity, psychiatry and general well-being. “The major challenge for the field is understanding how the function and regulation of clock genes contribute to health and diseases,” she says.
>k_subscribe_ad<>/k_subscribe_ad<Some of the first hints that a disrupted clock could lead to cancer came in 2001, when two teams of epidemiologists working with independent datasets came to the same conclusion: that women who regularly worked the night shift had an increased chance of developing breast cancer. Later studies established a link between graveyard shifts and cancers of the colon, prostate and endometrium — which prompted the World Health Organization’s International Agency for Research on Cancer in 2007 to designate night-shift work involving circadian disruption as a probable carcinogen.
Researchers now generally think that nocturnal light, by decreasing the body’s natural synthesis of the clock-regulating hormone melatonin, explains such a cancer link — which suggests that taking melatonin pills or modulating ambient lighting could help mitigate the risk among shift workers. But prospectively testing that idea would be difficult given the number of study subjects and years needed to detect a small number of cancers — and as yet, says Steven Hill, a circadian cancer researcher at Tulane University, “there are no published studies or active interventional studies” to that effect.
Cancer never sleeps
Logistically — and financially — it is far easier to do such trials for cancer treatment than for cancer prevention, and it’s here that many researchers are now focused. Earlier this year, for example, a team led by circadian biologist Satchidananda Panda at the Salk Institute for Biological Studies in La Jolla, California, described two novel drugs that target key clock components and kill several different types of cancer cells in a laboratory dish, as well as slow the growth of brain tumors in mice.
The drugs, by reawakening the circadian clock in cancer cells, seemed to block biological functions that tumors rely upon, including a garbage cleanup system that helps clear away toxic byproducts and a molecular pathway needed for fat synthesis. Importantly, the drugs had this therapeutic effect on tumors without causing any overt toxicity in the mice.
In fact, there’s even some data to suggest that resetting the clock in tumors could mitigate the side effects of cancer experienced elsewhere in the body. Cancer biologist Thales Papagiannakopoulos of New York University — his postdoc adviser called him “Chronos” given his Greek roots and academic interests — showed that lung tumor cells secrete a factor that travels through the blood to the liver, where it disrupts normal oscillatory patterns. Such effects, Papagiannakopoulos says, “could explain a lot of the symptoms of having cancer.”
For example, the liver is critical for draining blood from several organs, including the spleen, and altered liver rhythms could lead to splenic backups and congestion that shorten the lifespan of blood cells. This could account for common side effects of cancer, including anemia and leukopenia, the low counts of blood cells experienced by many cancer patients. And if something similar is happening in muscle tissues, circadian dysfunction could underpin cachexia, a devastating loss of body weight and strength that often afflicts people in the final stages of cancer.
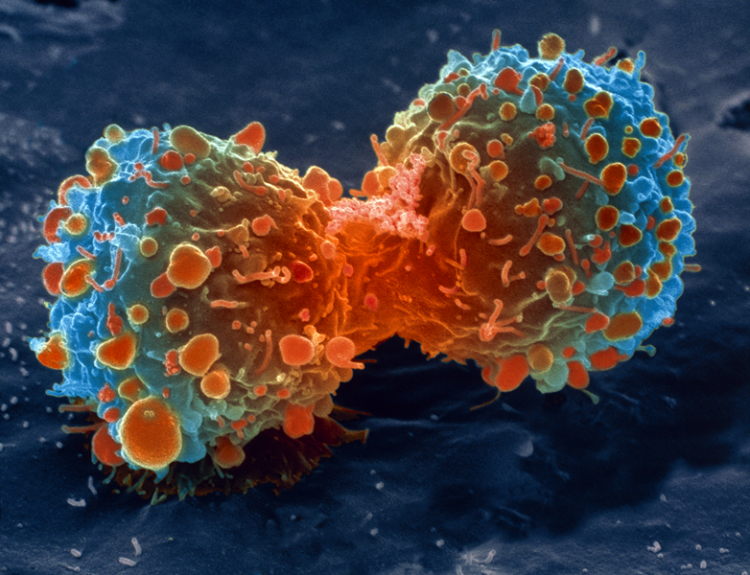
A colored scanning electron micrograph of a lung cancer cell undergoing division. Tumors often have malfunctioning circadian clocks.
CREDIT: SPL / SCIENCE SOURCE
Dang’s own therapeutics-related research has focused mainly on showing how a notorious cancer gene named MYC is involved in suppressing genes that lie at the very core of the mammalian clock, such as one called BMAL1. This perturbs the normal oscillatory cycles of molecular regulation inside the cell, and instead pushes protein synthesis into an aberrant, perpetual state of activity that drives tumor progression. This revealed a potential drug target — one that researchers at Texas Children’s Cancer Center have run with. Last year, they reported that a drug that indirectly stimulates BMAL1 activity could help blunt the growth of neuroblastoma, a cancer of the nerve tissue, both in cell cultures and in mouse models.
Dang, meanwhile, has recently turned his attention to a class of drugs that target NAMPT, an enzyme involved in both cancer metabolism and circadian feedback loops. Around 10 to 20 years ago, a handful of NAMPT inhibitors entered clinical testing. But they all caused such low platelet counts in recipients that they never progressed past early trials.
Now, working with mouse models of lymphoma, Dang’s team has found that administering these drugs at either 10 a.m. or 6 p.m. caused the same degree of tumor regression — with a key difference. Only at 6 p.m., when NAMPT expression was at its natural zenith in the liver, did the treatment cause low platelet counts in the mice. Inhibiting NAMPT at 10 a.m. caused no such problems whatsoever.
The work, which the group is retesting before submitting it for publication, has led Dang to think that chronotherapy could help salvage this promising class of new cancer drugs. “We may be interfering with liver function simply by giving those drugs at the wrong time,” he says.
Peaks and valleys
Another drug that doctors may currently be administering suboptimally is streptozocin, a chemotherapeutic agent that’s routinely used to treat a rare form of pancreatic cancer. In a paper published in 2017, Penn sleep medicine doctor Ron Anafi took reams of gene-activity data from both cancerous and healthy liver tissue, and then developed an algorithm that models all the molecular rhythms.
His analysis showed that activity levels of a gene called SLC2A2, which encodes a glucose transporter protein that shuttles streptozocin, cycled in a daily fashion in normal liver cells. Together with John Hogenesch, a chronobiologist at Cincinnati Children’s Hospital Medical Center, he then showed that administering streptozocin to mice at the nadir of SLC2A2 expression, when levels of streptozocin’s gatekeeper protein are low, minimized drug toxicity.
Anafi and Hogenesch hope to eventually test this time-optimized treatment schedule for streptozocin in patients with pancreatic tumors. At the moment, though, there is only one chronotherapy trial running in the United States (unlike in Europe and Asia, where these kinds of studies are more common). It’s happening at the Washington University School of Medicine in St. Louis, where neuro-oncologist Jian Campian and her colleagues have enrolled 30 people with brain cancer and given them a standard chemotherapy drug called temozolomide either at 8 a.m. or 8 p.m.
So far, says Campian, it looks as if the side effects of the drug are far worse when people take their pills in the morning. It’s too early to say anything conclusive, although animal studies from Campian’s collaborators, Erik Herzog and Joshua Rubin, suggest that the drug should work best when activity of the core clock regulator BMAL1 is highest in the tumor cells.
That would imply that the 8 p.m. dosing strategy will work better than the 8 a.m. one, at least on average. But, as Herzog points out, “people are different from each other.” We have something called a chronotype that, at its most basic level, determines whether someone is a morning person or an evening person, and that will probably affect individual responses to drugs like temozolomide.
Ideally, says Herzog, the timing of treatments would be finely tuned to a patient’s unique chronotype, and thus to the peaks and crests of their internal molecular activity. “This might end up being the ultimate personalized medicine,” he says.
But measuring something like BMAL1 activity in a tumor is no small task. It’s invasive (one has to obtain the tumor samples), expensive and unfeasible to do routinely, in real time and for many patients. In search of an easier way, a research team at Northwestern University has a 30-person pilot study looking for genomic indicators of clock status in blood and saliva.
Yet most clock researchers have settled on tracking cruder measures like skin temperature and wrist actigraphy to get a rough estimate of whether the body is in its activity or rest phase. Those are imperfect proxies, acknowledges Francis Lévi, a luminary in the field of chronotherapy from the University of Warwick in the United Kingdom. “We still need to identify better biomarkers to personalize chronotherapy.”
Even if there were an ideal biomarker, there’s still the problem of the medical system being designed around a standard working day. It’s not built to administer drugs at off hours — and there’s little financial incentive for change. So rather than bend the system to the needs of the patient, it might be easier to bend the circadian clock to the needs of the system. This may be possible simply by time-shifting eating, sleeping, exercise habits or ambient lighting to get a patient’s biological rhythms in sync with the business hours of a chemo ward.
Or perhaps the tumor’s clock itself could be reprogrammed. In a study published in 2017, Silke Kiessling, a circadian biologist who conducted the work as a postdoc at McGill University in Canada, showed that injecting tumors in mice with a steroid drug called dexamethasone reset genes like BMAL1 and induced rhythmicity in the cancer cells. Conceivably, that means a patient could take a shot of steroid a few hours before treatment so that, no matter when therapy is scheduled, that patient gets the benefit of chronotherapy without the inconvenience.
“This could be applicable for any kind of cancer you can reach with a needle,” says Kiessling, now at the Technical University of Munich in Germany. She is also thinking about making an aerosolized formulation for extending the strategy to lung cancer. “I’m more than confident that this could work, even in humans.”
Bedtime routine
For now, most of the human data come from anecdotal reports like those of Joe Kuna, who was diagnosed with metastatic colon cancer in 2014 and given two to five years to live. Four years on, the 61-year-old has beaten back a tennis-ball-size tumor in his colon and 15 lesions in his liver. Kuna, who runs a family-owned bowling alley in Johnsburg, Illinois, opted to undergo chronotherapy at the nearby Block Center for Integrative Cancer Treatment.
“We still need to identify better biomarkers to personalize chronotherapy.”
Francis LéviFor the better part of two years after his diagnosis, Kuna would arrive at the center every other Tuesday, usually some time between 1 p.m. and 3:30 p.m., for his dose of oxaliplatin. Since different anticancer drugs kill cells in different ways, “each drug has a window of time” when it works best, says oncologist Keith Block, medical director of the clinic in Skokie, Illinois — and oxaliplatin, according to Block, seems to work best in the afternoon.
Kuna would sit in the cubicle with the palm tree painted on the wall and eat his tuna fish sandwich while the intravenous medication dripped into his veins through a port implanted over his right nipple.
Another one of the drugs in Kuna’s chemotherapeutic cocktail, fluorouracil, is considered more of a nighttime agent — which meant Kuna had to take home a special pump, about the size of a paperback novel, that he could carry around in a fanny pack or leave on the mattress next to him in bed. The pump was programmed to kick in at 10 p.m., initially as a slow trickle. The flow would ramp up until 4 a.m. before dialing back down again and finishing up at 10 in the morning.
If you ask Kuna, this unusual drug regimen, plus the diet he followed and supplements he took as directed by the clinic, is the reason he’s alive today. “I truly believe it’s why I’m still here,” he says. And although a scan from January 2018 revealed two new cancerous spots in his liver, Kuna is confident that, with surgery and more chronotherapy, he could be cancer-free once more. Doctors removed the tumors on April 26.
But there’s no proof, without controlled trials, that the therapy as Kuna received it made a difference. Notably, of the other patients whom Kuna befriended at the Block Center, none is alive today. Most chronotherapy strategies now in place are based on a limited understanding of clock biology — which frustrates experts like Dang.
“We really want to provide the mechanistic basis of why you treat at a certain time of day,” Dang says, “and not just rely on trial and error.”
It’s defining why those little tweaks to common drug regimens improve patient outcomes that captures Dang’s imagination as he stares out the window of the Amtrak train on his semiweekly commute from Philadelphia, where he lives and runs his lab, to New York, for his office job at the Ludwig. Just as a small career move reshaped his own research agenda, “it could be,” he says, “that simple adjustments make a big difference for patients.”




